Diwakar Shukla
Unraveling the Molecular Magic of Witchweed
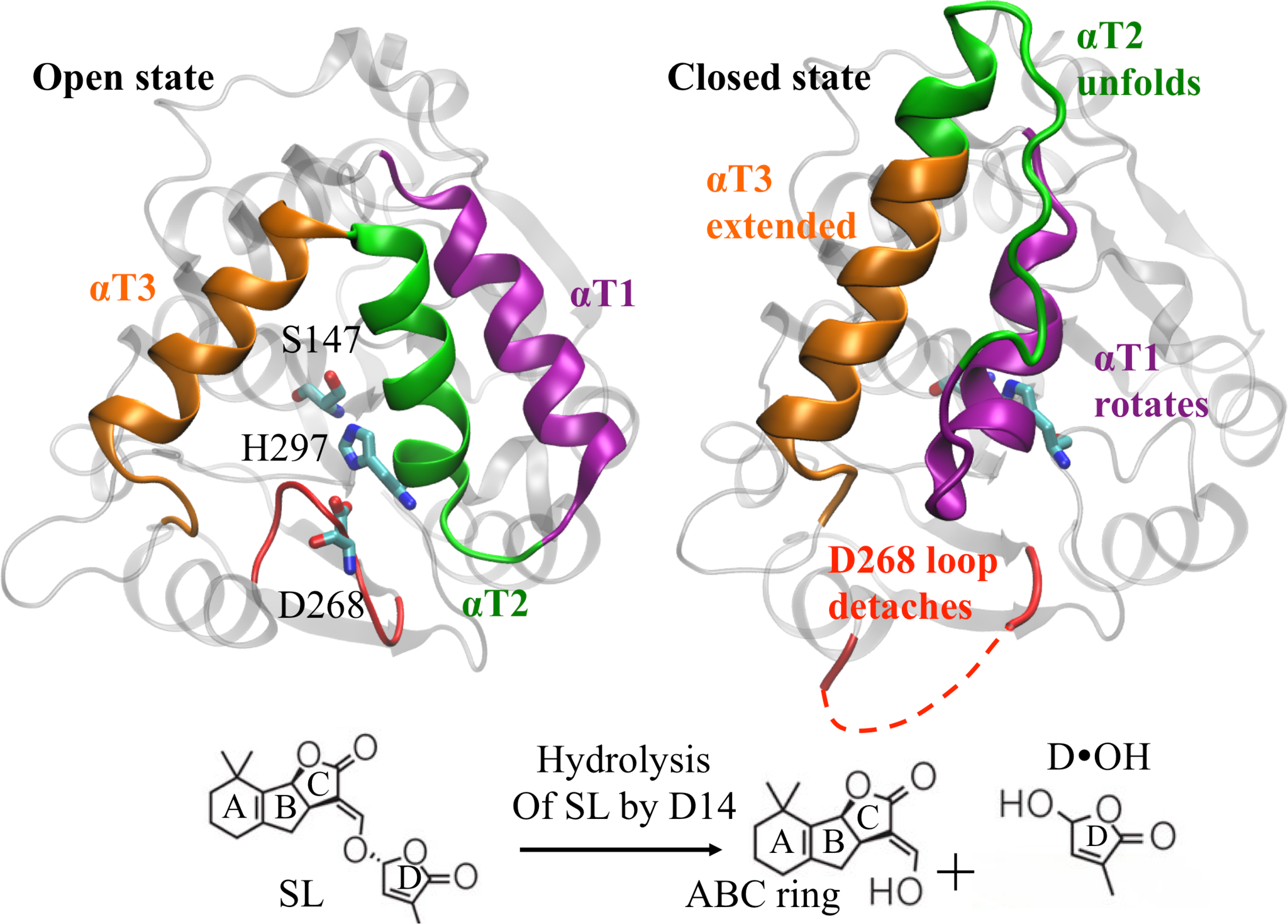
Witchweed, or Striga hermonthica, is a parasitic weed and an agricultural pest that destroys its host plants by using strigolactone (SL), a plant hormone released by the host plant into the soil, as a germination stimulant, after which it absorbs nutrients from its host and kills it. Weeds sense these hormones with pico-molar affinity whereas host plant proteins exhibit micro-molar affinity for these hormones. The origin of this affinity difference remains elusive as the host and parasite strigolactone receptors share high sequence structure and binding site conservation across species. This problem of ligand selectivity is central to the grand challenge of designing small molecules (herbicides) to target witchweed receptors with minimal effect on the host plant. Standard structural arguments cannot explain this selectivity. Therefore, this problem represents a critical challenge for agricultural productivity and future food security.
The central objective of Professor Shukla’s research project is to understand the molecular processes responsible for strigolactone selectivity in witchweed and employ the resulting hypothesis for the design of selective and potent inhibitors for witchweed. In particular, Professor Shukla and his research group will look at the molecular events involved in the strigolactone perception and subsequent receptor activation in atomistic detail using molecular simulations. They will employ the reinforcement learning based adaptive sampling methods they developed for conducting large-scale distributed simulations of these receptors. Finally,they will perform ligand design based on the intermediate states identified during the simulations.
An inhibitor for witchweed will help to reduce crop losses and improve food security in witchweed-vulnerable regions. Professor Shukla’s work approaches this design problem from a mechanistic standpoint and aims to gain insights into fundamental details currently inaccessible by experiments. Very little, if any, comprehensive mechanistic studies of SL signaling have previously been undertaken, so this work presents a novel synergistic integration of computational chemistry and plant biology approaches for addressing this critical challenge.
